2.4 Structure, Bonding, and Energy
Atomic Structure and Bonding Review
| bonding_revision.pptx | |
| File Size: | 165 kb |
| File Type: | pptx |
| electron_arrangements_ions_and_bonding_notes.pdf | |
| File Size: | 48 kb |
| File Type: | |
Chapter 9 - Properties of Solids
2.4.1 Particle Nature of Matter
|
Lesson Overview:
Most substances can occupy three states of matter: solid, liquid, and gas. The state they are in is determined by the strength of the attractive forces between particles in relation to the temperature (energy) and pressure. Particles behave differently in each state. This chapter will primarily be looking at how different types of solids bond, and the properties that result from those bonds.
|
| ||||||
2.4.2a Properties of Solids
|
Lesson Overview:
There are four different types of solids, each with their own characteristics and properties. Each will be discussed in more detail in their own sections below. -Ionic compounds are (usually) formed by the ionic bonding of a metal and a non-metal. -Molecular compounds are formed when non-metals bond covalently -Metals are the bonding of metal atoms -Network Covalent compounds are formed by large complex covalent networks of bonds and will be discussed individually as they have unique properties
|
The chart showing the relationship between different types of solids is at 6:18 below.
| ||||||
2.4.2b Ionic Solids
|
Lesson Overview:
Ionic compounds are formed by the ionic bonding of a positively charged cation and a negatively charged anion. Properties of ionic compounds: -High melting point -Brittle -Do not conduct a charge when solid -Conducts a charge when molten or in solution
|
| ||||||
2.4.2c Molecular (Covalent) Solids
|
Lesson Overview:
Molecular solids are formed by the covalent bonding of non-metals. Properties of molecular compounds include: -Low melting/boiling point -Brittle -Do not conduct electricity
|
| ||||||
2.4.2d Metallic Solids
|
Lesson Overview:
Pure metals are formed when only one type of atom is present. Alloys are formed when two or more types of metals are combined to form a metal with unique properties. Properties of metals include: -High melting/boiling points -Malleable and ductile -Good conductors of electricity
|
| ||||||
2.4.3 Network Solids
|
The first half of this video describes the structure of various silica compounds. We do NOT need to know the details of various semiconducting properties discussed in the second half.
| ||||||
Chapter 10 - Bonding
|
|
2.4.4 Inter- vs Intra- Molecular Forces
|
Three types of intermolecular forces that go by several names
1. Hydrogen Bonds are a specific type of polar bond. All hydrogen bonds are polar bonds but not all polar bonds are hydrogen bonds. 2. Polar bonds (aka. dipole-dipole interaction) are the attractive forces between polar molecules. 3. Molecular bonds (aka London dispersion or simply dispersion, and Van der Waals forces) are the weak attractive forces of non-polar molecules. These are often the result of induced dipoles.
|
| ||||||
| solids_and_bonding_3.ppt | |
| File Size: | 1776 kb |
| File Type: | ppt |
2.4.5 Lewis Structure Diagrams
|
| ||||||||||||||||||||||||||||||
2.4.6 Electronegativity and the Ionic-Covalent continium
2.4.7 VSEPR Theory
|
| ||||||
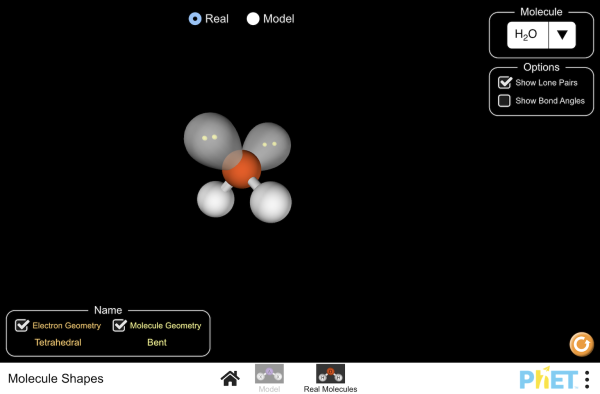
2.4.8 Molecular Geometry
|
| ||||||||||||
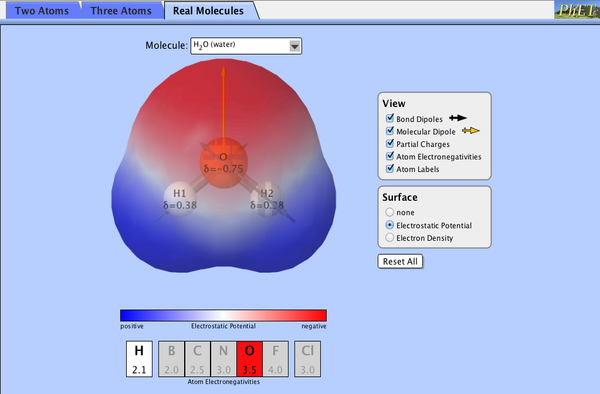
2.4.9 Polarity
|
| ||||||
2.4.10 Predicting Polarity of Molecules
|
| ||||||||||||
This is the demonstration that was done in class. If you want to watch it again without the american accent or hear polarity and solubility explained again.
2.4.11 Solubility
|
|
Chapter 11 - Energy Changes
|
|
2.4.12 Change in Enthalpy: Endo- vs Exo- Thermic Reactions
2.4.13 Activation Energy and Energy Diagrams
|
|
2.4.14 Calculating Heat of Reaction
|
| ||||||
2.4.15 Bond Energies
|
| ||||||
2.4.16 Thermochemical Equations
|
| ||||||||||||
Unit 2.4 Exam Review
(...in progress...)