2.0 Chemistry Warrant of Fitness
Chapters 1-3
| foundation_knowledge_pre-test_v2_answers.pdf | |
| File Size: | 1043 kb |
| File Type: | |
The purpose of this unit is to refresh your basic chemistry knowledge and skills. These key skills will be essential for success in Chemistry this year. If you are having trouble with any of these skills, please seek additional help from your teacher and get extra practice from the textbook and CD.
By the end of this unit you should be able to:
(See Chapter 1)
By the end of this unit you should be able to:
(See Chapter 1)
- State that matter is made up of particles, between which there is nothing but empty space.
- Define an element as a substance that cannot be broken down into simpler substances.
- State that compounds are made when atoms of different elements combine.
- Explain the difference between an oxygen atom and an oxygen molecule.
- Use chemical symbols correctly.
- Recall the mass, charge and location of electrons, protons and neutrons.
- Describe Rutherford’s model of the atom.
- Define the terms isotope, atomic number, mass number and ion.
- State the number of protons, neutrons and electrons in a given particle given its charge, atomic number and mass number.
- Write electron configurations for given atoms and ions from the first 20 elements of the periodic table.
- Name covalent and ionic compounds given their formulae, and write formulae for given compounds. (A table of ions will NOT be provided in exams.)
- Classify compounds as ionic or covalent from their names or formulae.
(See Chapter 2) - Recall the structure of the modern periodic table.
- Relate the similar chemical properties of a group of elements within the periodic table to their similar electron arrangements.
- Predict the electron arrangement of an element from its position in the periodic table and vice versa.
(See Chapter 3) - Recall general equations for common reactions.
- Recall the names and formulae of common laboratory reagents.
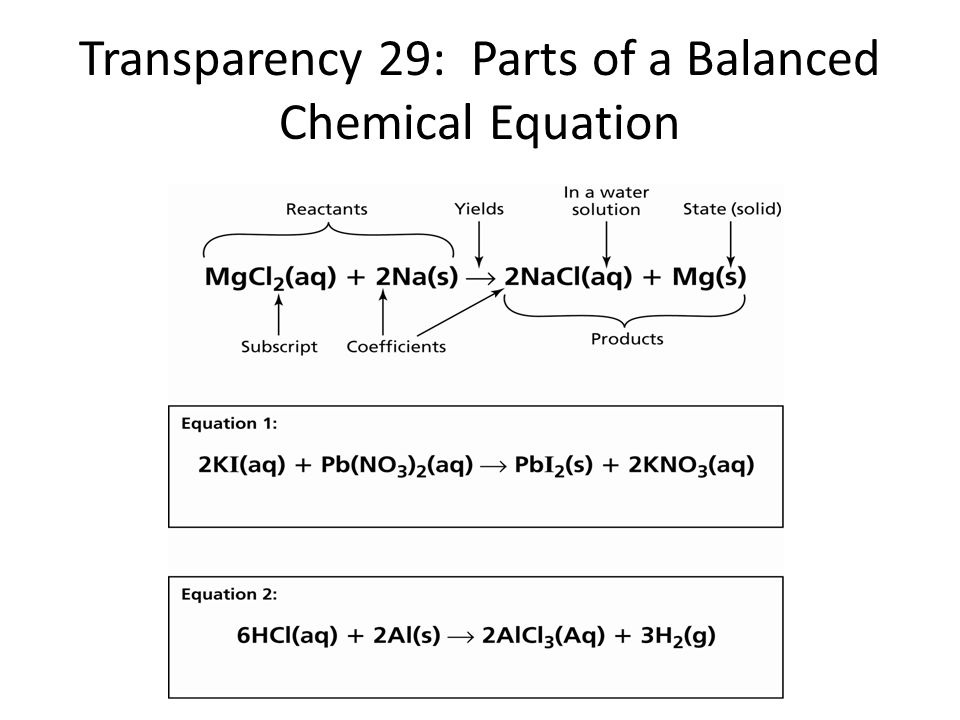
- Write balanced formula equations.
- For a given equation, state the number of each species present as reactant or product.
Chapter 1 - Atoms, Elements, Molecules and Compounds
2.0.1 Key Vocabulary and Structure of the Atom
|
HOMEWORK 2.0.1a: Text book questions 1B, 1C, 1D, 1E
HOMEWORK 2.0.1b: Text book Chapter 1 Review Questions Key Vocabulary: Use the Quizlet app below to study and test your knowledge
|
|
Chapter 2 - Chemical Families
2.0.2 Periodic Table
|
HOMEWORK 2.0.2: Textbook Questions
|
|
Chapter 3 - Equations in Chemistry
This chapter builds the foundation for equations in chemistry that we will use the rest of the year. If you are struggling with any of the concepts in each section, please seek additional help and do any of the optional homework for extra practice.
2.0.3 CHEMICAL FORMULA AND CHEMICAL EQUATIONS
|
HOMEWORK 2.0.3: Textbook Questions 3A
***More Practice (optional): CrissCross Ions Worksheet and Ion Formation Worksheet
Recommended Homework:
|
| ||||||||||||
2.0.4 Balancing Chemical Equations
|
HOMEWORK 2.0.4: Textbook Questions 3B
***More Practice (optional): Balancing Equations Worksheet
|
| ||||||
2.0.5 Maths in Chemistry
|
HOMEWORK 2.0.5 : Textbook Questions 3C
***More Practice (optional): Chapter Review Important Maths skills you should be comfortable with:
|
|