2.7 Reuction and oxidation
Introduction:
The Redox unit used to be an external standard and will be assessed in a written format. The following video provides an overview of redox chemistry. |
Learning Outcomes
| ||||||
2.7.1 Intro to Redox Reactons
|
Lesson Overview:
Understand that redox reactions involve the transfer of electrons from one atom or molecule to another. Elements have different properties when they have a different number of electrons (oxidation states), like the example of metal copper vs copper (II) ion in solution. |
|
2.7.2 Oxidation Numbers
|
Lesson Overview:
Recognise that the oxidation number is a representation of the number of electrons involved in a chemical bond. Calculate the oxidation number for atoms in common compounds. Definition:
Oxidation number, also called Oxidation State, the total number of electrons that an atom either gains or loses in order to form a chemical bond with another atom. Description: Each atom that participates in an oxidation-reduction reaction is assigned an oxidation number that reflects its ability to acquire, donate, or share electrons. The iron ion Fe3+, for example, has an oxidation number of +3 because it can acquire three electrons to form a chemical bond, while the oxygen ion O2− has an oxidation number of −2 because it can donate two electrons. In an electronically neutral substance, the sum of the oxidation numbers is zero; for example, inhematite (Fe2O3) the oxidation number of the two iron atoms (+6 in total) balances the oxidation number of the three oxygen atoms (−6). Certain elements assume the same oxidation number in different compounds; fluorine, for example, has the oxidation number −1 in all its compounds. Others, notably the nonmetals and the transition elements, can assume a variety of oxidation numbers; for example, nitrogen can have any oxidation number between −3 (as in ammonia, NH3) and +5 (as in nitric acid, HNO3). In the nomenclature of inorganic chemistry, the oxidation number of an element that may exist in more than one oxidation state is indicated by a roman numeral in parentheses after the name of the element--e.g., iron(II) chloride (FeCl2) and iron(III) chloride (FeCl3). (From Encyclopedia Britannica http://www.britannic_a.com/science/oxidation-number) HOMEWORK 2.7.2: Read Textbook pages 59-60 and answer all questions in 5C
Notes and worksheets with answers:
Homework: Worksheet "Oxidation Numbers and Redox"
|
| ||||||||||||
2.7.3 Using Oxidation States to determine REDOX
|
Lesson Overview:
Use Oxidation numbers to determine: (a) if a reaction is a redox reaction and if so, (b) which species were oxidised and reduced, and (c) what was the oxidising and reducing agents. Notes and worksheets with answers:
In Class Activity to be finished as homework if not completed in class: Worksheet "Oxidation States and Redox"
|
| ||||||||||||
2.7.4 Half reactions
|
Lesson Overview:
During a redox reaction, one species is reduced while another is oxidised. To make the reaction easier to understand, we can break the overall reaction into two halves, one for the oxidation and one for the reduction. A balanced "half reaction" can be written for each portion. In a later lesson we will learn how to combine them into a full balanced equation. Notes and Worksheet with answers
|
| ||||||||||||
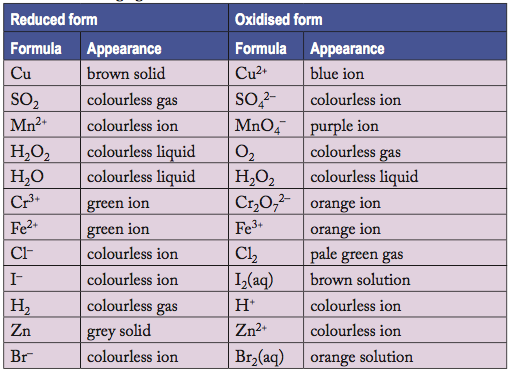
2.7.5 Colour Changes and other observations
|
Lesson Overview:
Many elements will present with different colours when in different oxidation states (see videos for examples). The colours can be used to help identify what has happened (what species are present) during a redox reaction. When colour can not be used as a sole means for identification, testing methods used in chapter 4 can still be used to identify common ions that may be present. In Class Activity: Practical 5.1
More practice writing half reactions and answers
Refer to the chart on Text page 56 for colours of species
|
Transition metals are well-known for their multiple colored ions. Watch the changes in color as manganese changes its oxidation state from +7 to +6 to +4.
| ||||||||||||
2.7.6 Balancing Redox Equations
|
Lesson Overview:
Bringing everything together. Once you have used reaction observations (or information provided) do identify species involved in a redox reaction, and balanced half reactions have been written, they can be combined to write a full balanced redox equation. Remember to always double check your work:-Are changes (electrons) balanced? -Are atoms balanced? -Do the change in oxidation states balance with the change in electrons? HOMEWORK Textbook Questions 5A and 5B
More Practice
Worksheet (two parts then answers) including balancing redox equations
Text Problems 5A
| |||||||||||||||||||
2.7.7 Hydrogen Peroxide
|
Lesson Overview:
Hydrogen peroxide has oxygen in a -1 oxidation state and can be either oxidized, reduced, or both. pH of the solution can have a large impact on the reactions of hydrogen peroxide. Detailed observations of any reaction will be very important in determining what sort of redox reaction is taking place. |
|
2.7.8 Other important redox reactions
|
In case you missed this demonstration during class.
|
|
2.7.9 Exam Prep
Key points to remember when answering questions for this exam
-List all observations (colours, states, etc.) that would be expected
-Link each colour/observation to a specific species
-Identify the oxidation number for the target atom in each relevant species
-Redox is about CHANGE; identify each key factor before AND after the reaction
-List all observations (colours, states, etc.) that would be expected
-Link each colour/observation to a specific species
-Identify the oxidation number for the target atom in each relevant species
-Redox is about CHANGE; identify each key factor before AND after the reaction
Previous exams and answers
|
| ||||||||||||
|
| ||||||||||||
|
| ||||||||||||